Biocontrol of Meloidogyne incognita (Kofoid and White) Chitwood, with the application of biological control agents
Abstract
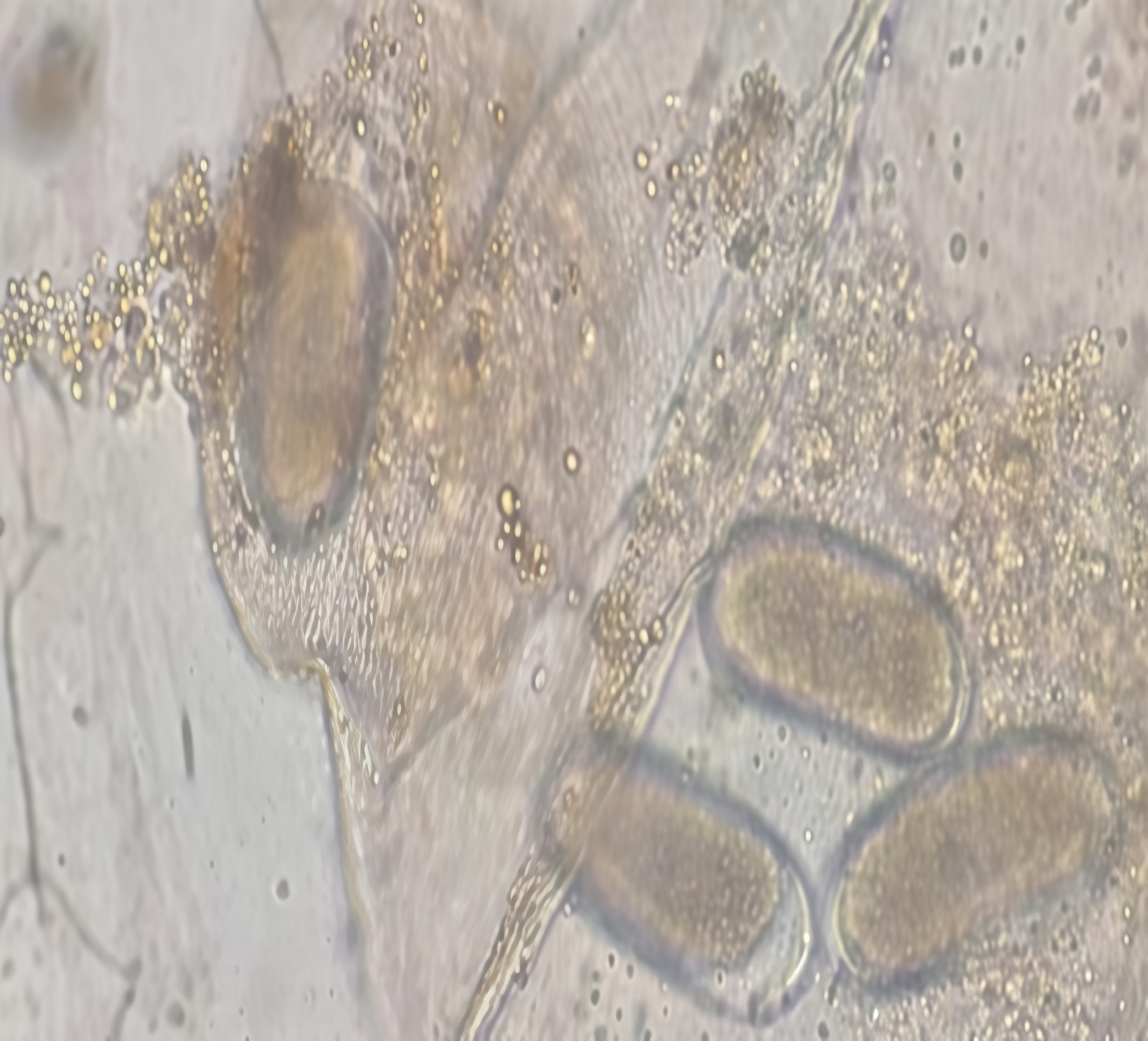
The objective of the research was to determine the potential of Trichoderma spp. strains, Isaria fumosorosea, and Isaria javanica as biocontrol agents against Meloidogyne incognita (Kofoid and White) Chitwood, obtained from tomato cv. Saladette (Solanum lycopersicum L.). Strains of T. harzianum, T. viride, T. koningii, T. asperellum, and a Trichoderma sp. isolate, as well as I. fumosorosea and I. javanica, were used. These were previously selected for their high parasitic capacity, antibiosis, and adaptation to diverse environmental conditions and substrates. In the in vitro assays, parasitism of the biological control agents (a pure filtrate of each strain) on eggs, oothecae, and juveniles (J2) of M. incognita was evaluated. Observations were carried out using an optical microscope with a 40X objective lens at 10 days and 72 hours, respectively. Nematodo control under semi-controlled conditions was conducted in an experimental area using 10 kg polyethylene bags, inoculated with approximately 5,000 juveniles (J2) and planted with tomato seedlings. Seven days after nematode inoculation, the biological agents were applied to the soil; 45 days later, incidence and severity variables were evaluated. Based on the results obtained, it was found that the strains of T. harzianum, T. asperellum, T. koningii, and I. fumosorosea are efficient for the control of different stages of the biological cycle of M. incognita.
Downloads
References
Ali, Q., Yu, C., Wang, Y., Sheng, T., Zhao, X., Wu, X., Jing, L., Gu, Q., Wu, H., & Gao, X. (2023). High killing rate of nematode and promotion of rice growth by synthetic volatiles from Bacillus strains due to enhanced oxidative stress response. Physiologia Plantarum, 175(1), e13868. https://doi.org/10.1111/ppl.13868
Ayaz, M., Zhao, J.T., Zhao, W., Chi, Y.K., Ali, Q., Ali, F., Khan, A.R., Yu, Q., Yu, J.W., Wu, W.C., Qi, R.D., & Huang, W.K. (2024). Biocontrol of plant parasitic nematodes by bacteria and fungi: A multi-omics approach for the exploration of novel nematicides in sustainable agriculture. Frontiers in Microbiology, 15, 1433716. https://doi.org/10.3389/fmicb.2024.1433716
Benedetti, T., Huzar-Novakowiski, J., Sordi, E., Carvalho, I.R., & Bortoluzzi, E.C. (2021). Microorganisms in the biological control of root-knot nematode: A meta-analytical study. Society and Development, 10(6), e39310615209. https://doi.org/10.33448/rsd-v10i6.15209
Benítez, T., Rincón, A.M., Limón, M.C., & Codón, A.C. (2004). Biocontrol mechanisms of Trichoderma strains. International Microbiology, 7(4), 249-260. https://scielo.isciii.es/pdf/im/v7n4/Benitez.pdf
Blanco Villacorta, M.W., Cadena Miranda, F.A., Challco Challco, G.J., & Lopez Loza, G. (2024). Efecto de Paecilomyces y Trichoderma spp. en el control biológico de nematodos en el cultivo de tomate (Solanum lycopersicum), en la Estación Experimental Patacamaya. Apthapi, 10(3), 2758-2766. https://doi.org/10.53287/pgiv2172jk63w
Calderón-Urrea, A., Vanholme, B., Vangestel, S., Kane, S.M., Bahaji, A., & Gheysen, G. (2016). Early development of the root-knot nematode Meloidogyne incognita. BMC Developmental Biology, 16, 10. https://doi.org/10.1186/s12861-016-0109-x
Di Rienzo, J. A., Casanoves, F., Balzarini, M. G., González, L., Tablada, M., & Robledo, C. W. (2017). InfoStat Profesional versión 2.1 [Software]. Grupo InfoStat. https://repositorio.catie.ac.cr/handle/11554/10346
Druzhinina, I.S., Seidl-Seiboth, V., Herrera-Estrella, A., Horwitz, B.A., Kenerley, C.M., Monte, E., Mukherjee, P.K., Zeilinger, S., Grigoriev, I.V., & Kubicek, C.P. (2011). Trichoderma: The genomics of opportunistic success. Nature Reviews Microbiology, 9(10), 749-759. https://doi.org/10.1038/nrmicro2637
Eisenback, J.D., Hirschmann, H., Sasser, J.N., & Triantaphyllou, A.C. (1981). A guide to the four most common species of root-knot nematodes (Meloidogyne spp.). North Carolina State University Graphics.
Harman, G.E. (2024). Integrated benefits to agriculture with Trichoderma and other endophytic or root-associated microbes. Microorganisms, 12(7), 1409. https://doi.org/10.3390/microorganisms12071409
Herrera-Parra, E., Ramos-Zapata, J., Cristóbal-Alejo, J., Tun-Suárez, J., & Reyes-Ramírez, A. (2018). Species of Trichoderma antagonistic to the root-knot nematode (Meloidogyne incognita) in habanera pepper. Phyton, 87, 7-13. https://www.revistaphyton.fund-romuloraggio.org.ar/vol87/v87a01_Herrera-Parra.pdf
Hooper, D.J., Hallmann, J., & Subbotin, S.A. (2005). Methods for extraction, processing and detection of plant and soil nematodes. In M. Luc, R.A. Sikora, & J. Bridge (Eds.), Plant Parasitic Nematodes in Subtropical and Tropical Agriculture (pp. 53-86). CABI. https://doi.org/10.1079/9780851997278.0053
Hussey, R.S., & Barker, K.R. (1973). A comparison of methods of collecting inocula of Meloidogyne spp., including a new technique. Plant Disease Reporter, 57, 1025-1028.
Kredics, L., Büchner, R., Balázs, D., Allaga, H., Kedves, O., Racić, G., Varga, A., Nagy, V.D., Vágvölgyi, C., & Sipos, G. (2024). Recent advances in the use of Trichoderma-containing multicomponent microbial inoculants for pathogen control and plant growth promotion. World Journal of Microbiology and Biotechnology, 40, 162. https://doi.org/10.1007/s11274-024-03965-5
Lewis, J., & Papavizas, G. (1983). Production of chlamydospores and conidia by Trichoderma spp. in liquid and solid growth media. Soil Biology and Biochemistry, 15(3), 351-357. https://doi.org/10.1016/0038-0717(83)90083-4
Martínez-Gallardo, J.Á., Díaz-Valdés, T., Allende-Molar, R., Retes-Manjarrez, J.E., & Carrillo-Fasio, J.A. (2019). Identificación y distribución de Meloidogyne spp. en tomate de Sinaloa, México. Revista Mexicana de Ciencias Agrícolas, 10(2), 453-459. https://doi.org/10.29312/remexca.v10i2.392
Migunova, V. D., & Sasanelli, N. (2021). Bacteria as biocontrol tool against phytoparasitic nematodes. Plants, 10(2), 389. https://doi.org/10.3390/plants10020389
Moo Koh, F.A., Cristóbal, J.A., Reyes, R.A., Tun Suárez, J.M., Gamboa, A.M., & Islas, R. (2018). Incompatibilidad interespecífica de especies de Trichoderma contra Meloidogyne incognita en Solanum lycopersicum. Scientia Fungorum, 47, 37-45. https://doi.org/10.33885/sf.2018.47.1191
Mukhtar, T., Tariq-Khan, M., & Aslam, M.N. (2021). Bioefficacy of Trichoderma species against javanese root-knot nematode, Meloidogyne javanica, in green gram. Gesunde Pflanzen, 73(3), 265-272. https://doi.org/10.1007/s10343-021-00544-8
Nicol, J.M., Turner, S.J., Coyne, D.L., de Nijs, L., Hockland, S., & Maafi, Z.T. (2011). Current Nematode Threats to World Agriculture. In J. Jones, G. Gheysen, & C. Fenoll (Eds.), Genomics and Molecular Genetics of Plant-nematode Interactions (pp. 21-43). Springer. https://doi.org/10.1007/978-94-007-0434-3_2
Ning, J., Zhou, J., Wang, H., Liu, Y., Ahmad, F., Feng, X., Fu, Y., Gu, X., & Zhao, L. (2022). Parallel evolution of C-type lectin domain gene family sizes in insect-vectored nematodes. Frontiers in Plant Science, 13, 856826. https://doi.org/10.3389/fpls.2022.856826
Sahebani, N., & Hadavi, N. (2008). Biological control of the root-knot nematode Meloidogyne javanica by Trichoderma harzianum. Soil Biology and Biochemistry, 40(8), 2016-2020. https://doi.org/10.1016/j.soilbio.2008.03.011
Sharon, E., Bar-Eyal, M., Chet, I., Herrera-Estrella, A., Kleifeld, O., & Spiegel, Y. (2001). Biological control of the root-knot nematode Meloidogyne javanica by Trichoderma harzianum. Phytopathology, 91(7), 687-693. https://doi.org/10.1094/PHYTO.2001.91.7.687
Sharon, E., Chet, I., Viterbo, A., Bar-Eyal, M., Nagan, H., Samuels, G. J., & Spiegel, Y. (2007). Parasitism of Trichoderma on Meloidogyne javanica and role of the gelatinous matrix. European Journal of Plant Pathology, 118(3), 247-258. https://doi.org/10.1007/s10658-007-9140-x
Servicio de Información Agroalimentaria y Pesquera [SIAP]. (2024). Producción agrícola. https://www.gob.mx/siap
Siddiqui, Z.A., & Mahmood, I. (1999). Role of bacteria in the management of plant parasitic nematodes: A review. Bioresource Technology, 69, 167-179. https://doi.org/10.1016/S0960-8524(98)00122-9
Sikora, R. A., Coyne, D., & Quénéhervé, P. (2018). Nematode parasites of bananas and plantains. In R.A. Sikora, D. Coyne, J. Hallmann, & P. Timper (Eds.), Plant Parasitic Nematodes in Subtropical and Tropical Agriculture (pp. 617-657). CABI. https://doi.org/10.1079/9781786391247.0617
Subedi, S., Thapa, B., & Shrestha, J. (2020). Overview of root-knot nematode (Meloidogyne incognita) and control management. Journal of Agriculture and Natural Resources, 3(2), 21-31. https://doi.org/10.3126/janr.v3i2.32298
Taylor, A.L., & Sasser, J.N. (1978). Biology, identification and control of root-knot nematodes (Meloidogyne species). North Carolina State University. https://www.cabidigitallibrary.org/doi/full/10.5555/19780844105
Vrain, T.C. (1977). A technique for the collection of larvae of Meloidogyne spp. and a comparison of eggs and larvae as inocula. Journal of Nematology, 9(3), 249-251. https://pmc.ncbi.nlm.nih.gov/articles/PMC2620246/
Copyright (c) 2026 Jesús Orlando Pérez-González, Humberto Rafael Bravo-Delgado, Yonger Tamayo Aguilar, Adolfo Amador Mendoza, Jorge Francisco León de la Rocha

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.



















